 |
The use of colorants as additives for food and drinks is a significant factor to food manufacturer and consumer alike in determining the acceptability of processed food. For manufacturer, added colorants assist in ensuring batch-to-batch uniformity and help reinforce colorants that are already present but are less intensive that the consumer would expect.
For the consumer, added colorant help to restore the original appearance of foods whose natural colorant content as been reduced by processing treatments also to provide appealing and readily identifiable products. In practice consumers do not isolate their sensory perceptions of a food but combine them in obtaining a total assessment for a food. This association is sufficiently strong that when asked to separate them there may still be a strong influence of one sensory perception on another. Yet the visual sense is both sophisticated and sensitive, it is only that experience his confirmed what the eye sees, which has caused the interaction of the senses to become unconscious.
Colouring fresh food is not permitted as a matter of principle. As a rule, colouring is used only for processed food with no colour of its own or in which residual amounts of colour remain. Food must not be coloured to simulate a higher level of nutritionally important components or worse, to mask poor quality or spoilage.
Colorant is generally added to food at very early stage in processing. Colorant agents must therefore be stable on heating, cooling, acid, or oxygen. In particular, they must remain stable during storage of the food when they are often exposed to light. Many natural colorants lack this stability so that, despite other advantages, their use is limited. Sulfur dioxide, which is used in a number of foods, can destroy many colours.
The use of colorants in food in the European Community (EC) is controlled by EC Directive 2645/62 (as amended) and in the USA by the Colour Additives Amendments of 1960 (Public Law 86-618) to the food Drug and Cosmetic Act 1938. These statutes make no provision for the terms "natural", "artificial" and "nature identical" although these descriptions are finding increasing use in the labelling and advertising of foods.
Artificial colorants, i.e. those commonly referred to as "coal tar dyes" or "food dyes", have for many years been ideal for replacing natural colorant destroyed during food processing, reducing colour variations in product batches and providing consumers appeal. However in recent years the number of dyes suitable for food use has been drastically reduced as a result of toxicological studies and, in Europe, by harmonization of European Community legislation. In addition there as been consumer pressure via major supermarket chains for more natural food products incorporating natural ingredients.
Natural colorants as a rule are more expensive, less stable, and possess lower tinctorial power. In addition, their are frequently present as mixtures in the host materials and vary with season, region, variety, etc. Disadvantages associated with natural colorants have been identified at low yields; colour instability resulting from effects of pH, light, heat, and freezing; and possible action with other properties that may be undesirable (Blenford, 1985). However, they do present the advantage of being perceived as safer than synthetic colours.
Pigments to be used as colorants must be extracted from the host material and prepared in some form, such as dried powdered beadlets, which increase the ease of use in a food product.
The breakthrough in use of natural pigments occurred with the synthesizing of the carotenoids b-carotene, b-apo-8'-carotenal, and canthaxanthin. A synthetically pure pigment can also be modified or prepared into a suitable market form to enhance its stability and to meet the wide variety of applications required by food manufacturers. One obvious success in this area has been the production of b-carotene forms that are pH stable, resistant to chemical reduction, and water dispersible.
Commercial exploitation of these procedures is unlikely to develop further because yields are usually low, resulting in high-cost additives; in addition, natural-identical colorants have to be confirmed as toxicologically safe before they are adopted by regulatory authorities, and then this only occurs if there is perceived need.
Manufacturers of natural colorants have concentrated their efforts on maximizing yields from conventional sources. Extraction methods usually involve disruption of the plant material followed by acidified aqueous extraction for anthocyanins, beetroot red, and cochineal or by solvent extraction for chlorophyll, carotenoids, annatto and spice extracts. Stabilizers such as citric acid, ascorbic acid and antioxidants help reduce pigment loss during extraction.
Selection of a suitable cultivar of plant is a cost-effective way of improving pigment yield. Plant tissue culture will, in the future, assist suppliers in selecting highly pigmented cultivars.
Enzymes offer a mean of enhancing natural colorant extraction but their use has been limited, presumably by the low value of pigments. Pectinase and cellulases have, however, been used to produce high carotene content in carrot juice. Bromelain and b-glucosidase, too, have been used in extracting pigments from gardenia fruits, while protease treatment of Dactylopius coccus produces a better quality of cochineal.
Natural colorant extracts, in particular those produced by aqueous extraction from or direct pressing of plant materials, contain a high proportion of other dissolved solids including salts, organic acids, phenolic compounds and sugars. Drying these high-solid extracts produces hygroscope powders, which have certain drawbacks. In the case of beetroot, the powder has a characteristic odour (geosmin) and high nitrate and nitrite content and limited application in foods. In anthocyanin extracts, coextracted sugars may degrade to furfural and 5-hydroxy- methyl-furfural during dehydration and contribute to accelerated pigment degradation. The problem can be minimized by treating the extracts with microorganisms.
Approximately 80% of beetroot juice solids are carbohydrate and nitrogenous compounds. Fermentation of this juice using Candida utilis under partial anaerobic conditions reduced solids substantially, giving five- to sevenfold increase in pigment content on a dry weight basis (Adams et. al., 1976). In addition, the dried product lacked the characteristic beetroot odour and had a reduced nitrate content. Yeast cells produced during the fermentation, which takes about seven hours at 30�C, may be harvested by centrifugation and used as a source of biomass for animal feed supplement.
Another approach to reducing the content of co-extractants during pigment purification is the use of supercritical carbon dioxide as an extracting solvent. Some success has been obtained in extracting carotene, bixin and chlorophyll from natural sources.
Membrane processing has found wide application in concentrating and separating food ingredients of a thermally labile nature. Three main overlapping operations can be defined according to the size of molecules or particles that can be retained and the operating pressures used:For natural colouring production, membrane offers a number of benefits including concentration of pigments from dilute juices; removal of microorganisms from fermented or infected juices and removal of cell debris from plant extracts. Ultrafiltration of anthocyanin- and betanin-containing extracts using cellulose acetate membranes with molecular weight cut- off of 1000 Da and 500 Da, respectively has been shown to remove 75-90% of water and 50- 60% of sugars in a single pass. This represents a twofold increase in pigment content and thus a viable extraction method.
Plant cell culture in fermentors rather than fields has potential for the production of secondary metabolites such as pharmaceuticals, perfumes, flavours and colourings. Commercial success has been achieved with shikonin, a red naphtoquinone dye produced in Japan and used to promote healing. The potential of plant cell cultures has resulted in the development of the technique from relatively low-yielding heterogeneous cell suspension, through immobilization technique and the use of "hairy root" cultures.
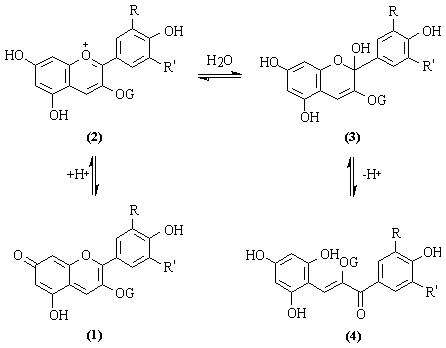
In a chemical point of view there are basically two categories of colour compounds: conjugated polyenes and metalloporphyrins. The former includes carotenoids, annatto, anthocyanins, betanain, dyes and lakes. The effect of conjugation is to lower the p-p* transition energy from the highest occupied molecular orbital. Increased conjugation in the molecule shifts the absorption maximum to a higher wavelength. Substituint groups with lone pairs of electrons tend to increase p conjugation by resonance.
 |
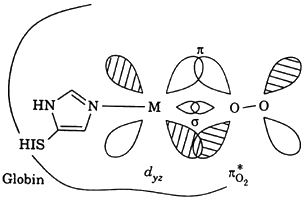
The two best-known examples of metalloporphyrins found in food are the myoglobin and chlorophylls. A porphyrin metal complex possesses 19 p-electron in an 18-atom ring. The main effect of the metal on the transitions is the conjugation of the metal p orbital with the porphyrin p orbital. The splitting of d orbital of the metal ion because of the porphyrin results in additional loss of degeneracy from the theoretically predicted octahedral symmetry.
 |
The most important colorants commonly used in foods are summarized bellow. In contrast to other food additives, major differences exist in the food-colouring laws of the EEC countries, Japan, and USA.
| EEC n� | Natural | Name | Colour (solvent)1 | Formula |
|---|---|---|---|---|
| E 100 | yes | curcumin | Yellow-reddish (E) | view |
| E 101 | yes | riboflavin (lactoflavin) | Yellow (W) | |
| E 102 | no | tartrazine | Lemon-yellow (W) | view |
| E 104 | no | quinoline yellow | ||
| E 110 | no | sunset yellow FCF | Orange (W) | view |
| E 120 | yes | cochineal (carminic acid) | Bright-red | view |
| E 122 | no | carmoisin (azo rubine) | Red with bluish tint (W) | view |
| E 123 | no | amaranth | Red with bluish tint (W) | view |
| E 124 | no | ponceu 4R (cochineal red A) | Scarlet-red (W) | view |
| E 127 | no | Erythrosin | Cherry-red (W) | view |
| E 131 | no | patent blue V | Blue with a greenish tint (W) | view |
| E 132 | no | indigo carmine (indigotine) | Purple blue (W) | view |
| E 140 | yes | chlorophyll | Green | view |
| E 141 | no | chlorophyll copper complexes | Green | |
| E 142 | no | green S (acid brilliant BS or lissamine green) | Green (W) | view |
| E 150 | no | caramel | ||
| E 151 | no | black PN (brilliant black BN) | Violet with bluish tint | view |
| E 153 | no | carbon black (vegetable carbon) | ||
| E 160 | yes | carotenoids (carotene, annatto, bixin, norbixin, capsanthin, capsorubin, lycopene, b-apo-8'-carotenal and its ethyl ester) | Orange (oil) | |
| E 161 a-g | yes | flavoxanthin, lutein, cryptoxanthin, rubinxanthin, violaxanthin, rhodoxanthin, canthaxanthin | Orange (oil) | |
| E 162 | yes | beetroot red (betanin) | ||
| E 163 | yes | anthocyanins | Red-violet2 (W) | view |
| E 171 | no | titanium dioxide | ||
| E 172 | no | iron oxides and hydroxides | ||
| E173-E175 | no | metals (aluminum, silver, gold) | ||
| E 180 | no | pigment rubine (lithol rubine BK) |