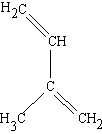
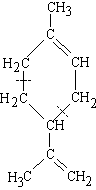
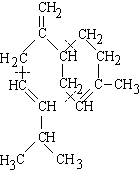
(2-methyl-1,3-butadiene)
Terpenes
Terpenes are the most common hydrocarbons found in the essential oil of dry brown plants.
A wide variety of cyclic terpene hydrocarbon are known and, as multiples of (C5H8), these have fewer double bonds than the open-chain terpenes.
The chemical structure of the terpenes was established by Wallach in 1887, by proposing the so-called "isoprene rule", by which the terpenes are considered to be fusions of two or more isoprene (C5H8) unities. The general formula of terpenes is (C5H8)n. If n=2 the (C10) compounds are called monoterpenes; if n=3 they are known as sesquiterpenes and when n=4 as diterpenes. Some triterpenes (C30) and tetraterpenes (C40) are also known, as, for example, the squalene, an acyclic triterpene (C30H50) and the lycopene, an acyclic tetraterpene (C40H56), or the bicyclic tetraterpene, the b-carotene.
 |
 |
 |
| isoprene (C5H8) (2-methyl-1,3-butadiene) |
dipentene (C10H16) | cadinene (C15H24) |
| Monoterpenes | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Acyclic | ocimene myrcene |
| |||||||||
| Monocyclic | dipentene iso-limonene limonene ( d and l ) a-phellandrene b-phelandrene ( d and l ) sylvestrene a-terpinene b-terpinene g-terpinene terpinolene |
| |||||||||
| Bicyclic | camphene ( d and l ) D3-carene ( d and l ) a-pinene ( d, l and dl ) b-pinene ( d and l ) sabinene ( d and dl ) |
| |||||||||
| Sesquiterpenes | ||||||
|---|---|---|---|---|---|---|
| Acyclic | a-farnesene | 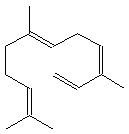 a-farnesene | ||||
| Monocyclic | a-bisabolene b-bisabolene g-bisabolene zingiberene |
| ||||
| Bicyclic | b-cadinene b-caryophyllene a-selinene |
|
| Diterpenes | |
|---|---|
| camphorene |  camphorene |
Ruzika (1959) developed the terpenoid theory which predicts that the biogenesis of terpenes probably takes place in four phases: the formation of the C5 precursor units; the linking together to form the primary terpenoid structure; cyclization; and specific rearrangements involving oxidation, reduction, hydroxylation.